A new type of battery developed at Oregon State University is the first to use only hydronium as its charge carrier. Although the technology is in very early stages, the team at OSU says that it could provide a more sustainable option for researchers, particularly in the area of utility-scale storage.
Energy storage is growing into an enormous market worldwide, and storage at utility scale is expected to be hugely popular in the U.S. Although lithium-ion is by far the dominant technology, and is expected to remain so, other chemistries are able to get a look in at some points, including in U.S utility scale.
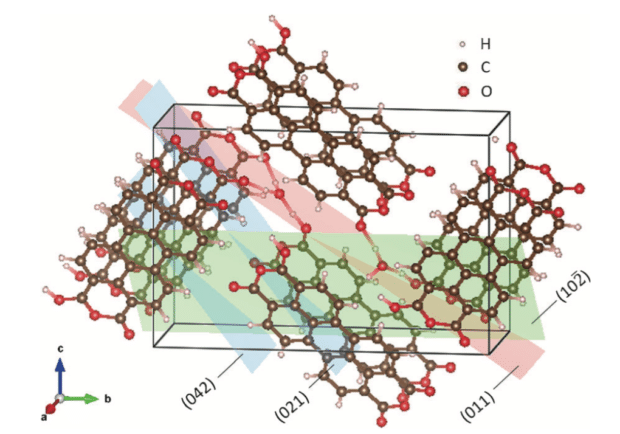
Researchers at OSU have demonstrated that hydronium ions can be reversibly stored in organic crystalline material PTCDA (perylenetetracarboxylic dianhydridem). The battery uses dilute sulfuric acid as an electrolyte.
“This may prove a paradigm-shifting opportunity for more sustainable batteries,” said OSU Assistant Professor of Chemistry Xiulei Ji. “It doesn’t use lithium, sodium or potassium to carry the charge, and just uses acid as the electrolyte. There’s a huge natural abundance of acid so it is highly renewable and sustainable.”
The majority of batteries available use alkali metals such as lithium to carry their charge, and another metal as an electrode. OSU’s Ji also noted that the combination of hydronium and PTCDA has the potential for high power and good capacity. “It’s not going to power electric cars,” he continues. “But it does provide an opportunity for battery researchers to go in a new direction as they look for new alternatives, particularly for stationary grid storage.”
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.







By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.